
New data presented March 22, 2017 at the Orthopedic Research Society Annual Meeting highlights the superior lubricating properties of HYMOVIS® against all other competitor HA products evaluated.
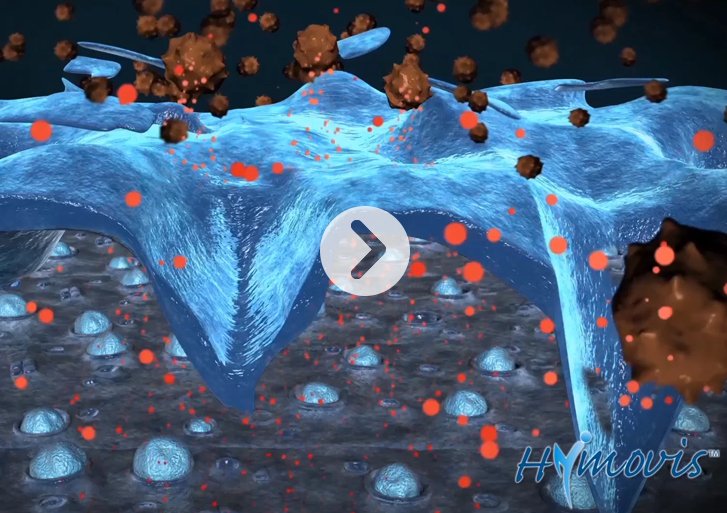
HYMOVIS® has exceptional lubricating properties in moving cartilage to a low-friction regime. The study provides a unifying framework to evaluate the potential clinical efficacy of injectable hyaluronic acid therapies.
Fidia Farmaceutici S.p.A., a world leader in the research, development and manufacturing of hyaluronic acid (HA) - based products and its wholly owned subsidiary, Fidia Pharma USA Inc., announces new data were presented at the Annual Meeting of the Orthopaedic Research Society (ORS) in San Diego, CA, March 19-22, 2017.
Scientists from Fidia Farmaceutici S.p.A. and the research group led by Dr. Lawrence Bonassar, Professor at the Meinig School of Biomedical Engineering at Cornell University evaluated the in vitro rheological and tribological properties of six commercial HA products used for the treatment of pain in osteoarthritis (OA) of the knee. “In our research, we demonstrated that there are clear differences in the lubricating abilities of this cohort of HA products. HYMOVIS® is an exceptional viscous lubricant in moving cartilage to a low-friction regime, having superior biomechanical lubricating properties amongst all the HAs evaluated.” In addition, the study approach offers a novel method to evaluate the potential clinical efficacy of injectable hyaluronic acid therapies that may be more sensitive than rheological measurements alone.
This new data were presented in the late breaking session of the Annual Meeting of the Orthopaedic Research Society (ORS) on Wednesday, March 22, 2017 (http://www.ors.org/Transactions/63/2374.pdf). (Pending publication).
HYMOVIS® is a highly viscoelastic, non-crosslinked hydrogel bioengineered using a proprietary process that increases lubrication and shock absorption properties. This results in a natural hyaluronan similar to the hyaluronan found in the synovial fluid present in human joints. The formulation allows this unique molecule to recover its original structure, even after repetitive mechanical stress. Due to reversible hydrophobic interactions, the non-crosslinked HYMOVIS® has increased elasticity, viscosity and residence time in the joint. Its unique molecular structure results in enhanced biomechanical properties and long-lasting efficacy, all in a convenient two-dose regimen.

|
Fidia offers COMPREHENSIVE SUPPORT for your patients and your practice The HYMOVIS Support Hotline*: 1-866-HYMOVIS (1-866-496-6847) or www.HYMOVISonline.com
|
| No minimum order quantity | |
| Low thresholds for contract pricing | |
| Easy online ordering | |
| Each carton contains two blister packs, each with a single-use syringe | |
| 3.0 mL of HYMOVIS® in a 5.0-mL single-use syringe | |
| A hyaluronan concentration of 8 mg/mL, dissolved in physiologic saline | |
 |
| J-Code: J7322 | |
| CPT 20610; 20611 physician administration | |
 |
| Intra-articular hyaluronans are covered by most insurance plans | |
| For reimbursement assistance, please call 1-866-HYMOVIS (1-866-496-6847) | |
| For patient insurance verification on the web register/login to: www.fidiacomplete.com | |
| Reimbursement Guide | |
| Prior Authorization Checklist 2017 | |
| Tips for Clean Claims Submission | |
| Patient Enrollment Form | |
| Coding information for HYMOVIS | |
| Overview of Reimbursement Support | |
| Samples CMS 1450 Form for HYMOVIS | |
| Samples CMS 1500 Form for HYMOVIS | |
| Strategies to Appeal Denied Claims |
| Available 9 AM to 8 PM ET, Monday through Friday | |||||||||
| Hotline: 1-866-HYMOVIS (1-866-496-6847) | |||||||||
Dedicated reimbursement support including:
|
|||||||||
| Simple ordering process | |||||||||
| Report adverse event or product complaints | |||||||||
| For Customer Service, please email us at customerservice@fidiapharma.us | |||||||||
| For product-related specific information, please contact the Medical Office at medicaloffice@fidiapharma.us | |||||||||
| For additional information please email us at hymovis@fidiapharma.us |
Fidia offers COMPREHENSIVE SUPPORT
for your patients and your practice
The HYMOVIS Support Hotline*: 1-866-HYMOVIS (1-866-496-6847)
or www.HYMOVISonline.com
| • | Available 9 AM to 8 PM ET, Monday through Friday | ||||||||||
| • | Dedicated reimbursement support | ||||||||||
| • | Three ways to enroll: phone, fax, or web
|
||||||||||
| • | Simplified ordering, billing, and return processing | ||||||||||
| • | Report adverse events or a product quality complaint | ||||||||||
| • | Contact sales representative to order posters or brochures | ||||||||||
| • | Medical information |
| * | The Hotline does not file claims for callers, nor can it guarantee that you will be successful in obtaining reimbursement. Third-party payment for medical products and services is affected by numerous factors, not all of which can be anticipated or solved by the Hotline. |
FIDIA offers
|
Fidia offers COMPREHENSIVE SUPPORT for your patients and your practice The HYMOVIS Support Hotline*: 1-866-HYMOVIS (1-866-496-6847) or www.HYMOVISonline.com
|
| No minimum order quantity | |
| Low thresholds for contract pricing | |
| Easy online ordering | |
| Each carton contains two blister packs, each with a single-use syringe | |
| 3.0 mL of HYMOVIS® in a 5.0-mL single-use syringe | |
| A hyaluronan concentration of 8 mg/mL, dissolved in physiologic saline | |
 |
| J-Code: J7322 | |
| CPT 20610; 20611 physician administration | |
 |
| Intra-articular hyaluronans are covered by most insurance plans | |
| For reimbursement assistance, please call 1-866-HYMOVIS (1-866-496-6847) | |
| For patient insurance verification on the web register/login to: www.fidiacomplete.com | |
| Reimbursement Guide | |
| Prior Authorization Checklist 2017 | |
| Tips for Clean Claims Submission | |
| Patient Enrollment Form | |
| Coding information for HYMOVIS | |
| Overview of Reimbursement Support | |
| Samples CMS 1450 Form for HYMOVIS | |
| Samples CMS 1500 Form for HYMOVIS | |
| Strategies to Appeal Denied Claims |
| Available 9 AM to 8 PM ET, Monday through Friday | |||||||||
| Hotline: 1-866-HYMOVIS (1-866-496-6847) | |||||||||
Dedicated reimbursement support including:
|
|||||||||
| Simple ordering process | |||||||||
| Report adverse event or product complaints | |||||||||
| For Customer Service, please email us at customerservice@fidiapharma.us | |||||||||
| For product-related specific information, please contact the Medical Office at medicaloffice@fidiapharma.us | |||||||||
| For additional information please email us at hymovis@fidiapharma.us |
*Mechanical testing may not be indicative of human clinical outcomes
** From a multi-center open label study in 49 patients treated with HYMOVIS®